
Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti‐Markovnikov‐Selective Hydrothiolation of Olefins - Sinha - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library

Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti‐Markovnikov‐Selective Hydrothiolation of Olefins - Sinha - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library
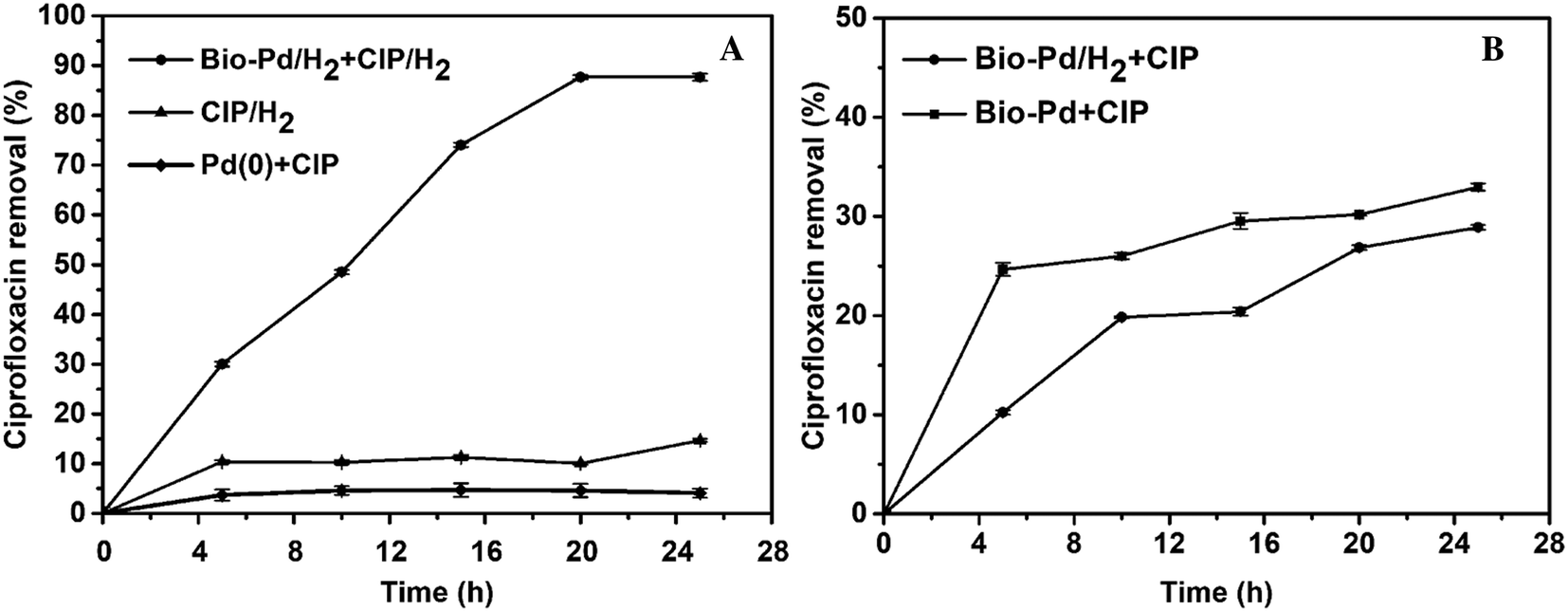
Enhanced reductive removal of ciprofloxacin in pharmaceutical wastewater using biogenic palladium nanoparticles by bubbling H 2 - RSC Advances (RSC Publishing) DOI:10.1039/D0RA03783D
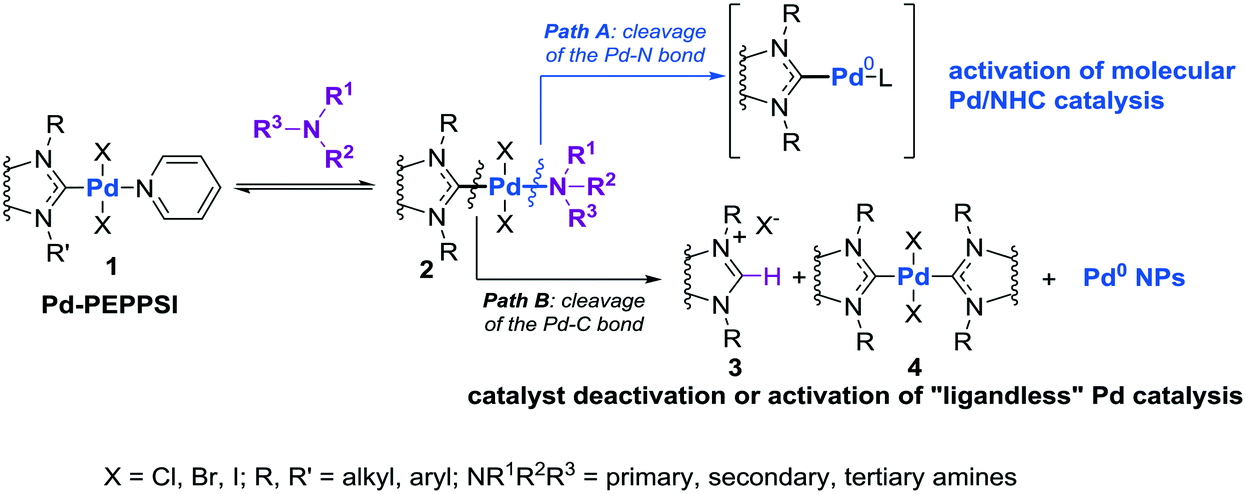
Preventing Pd–NHC bond cleavage and switching from nano-scale to molecular catalytic systems: amines and temperature as catalyst activators - Catalysis Science & Technology (RSC Publishing)
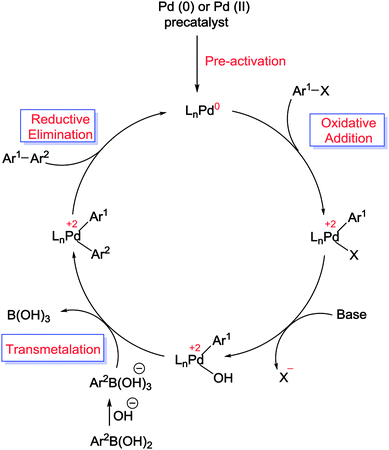
Applications of SBA-15 supported Pd metal catalysts as nanoreactors in C–C coupling reactions - RSC Advances (RSC Publishing)

Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect

TEM images of the thiol derivatized Au nanoparticles. The particle size... | Download Scientific Diagram
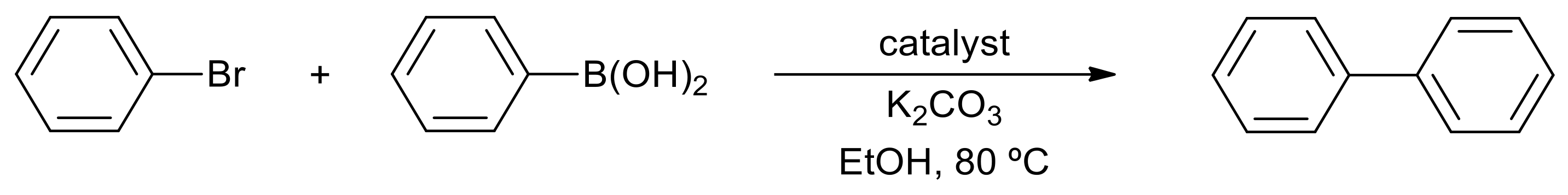
Materials | Free Full-Text | Thiol-Functionalized Ethylene Periodic Mesoporous Organosilica as an Efficient Scavenger for Palladium: Confirming the Homogeneous Character of the Suzuki Reaction

Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti‐Markovnikov‐Selective Hydrothiolation of Olefins - Sinha - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library
Engineering of Surface Environment of Pd Nanoparticle Catalysts on Carbon Support with Pyrene–Thiol Ligands for Semihydrogenat

Palladium‐Catalyzed C–S Bond Formation: Rate and Mechanism of the Coupling of Aryl or Vinyl Halides with a Thiol Derived from a Cysteine - Moreau - 2005 - European Journal of Organic Chemistry -

Thiol–enes: Chemistry of the past with promise for the future - Hoyle - 2004 - Journal of Polymer Science Part A: Polymer Chemistry - Wiley Online Library
Thiolâ‹™Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Antiâ•'Markovnikovâ•'Selective Hydrothiolat
Tuning the Catalytic Activity and Selectivity of Pd Nanoparticles Using Ligand-Modified Supports and Surfaces
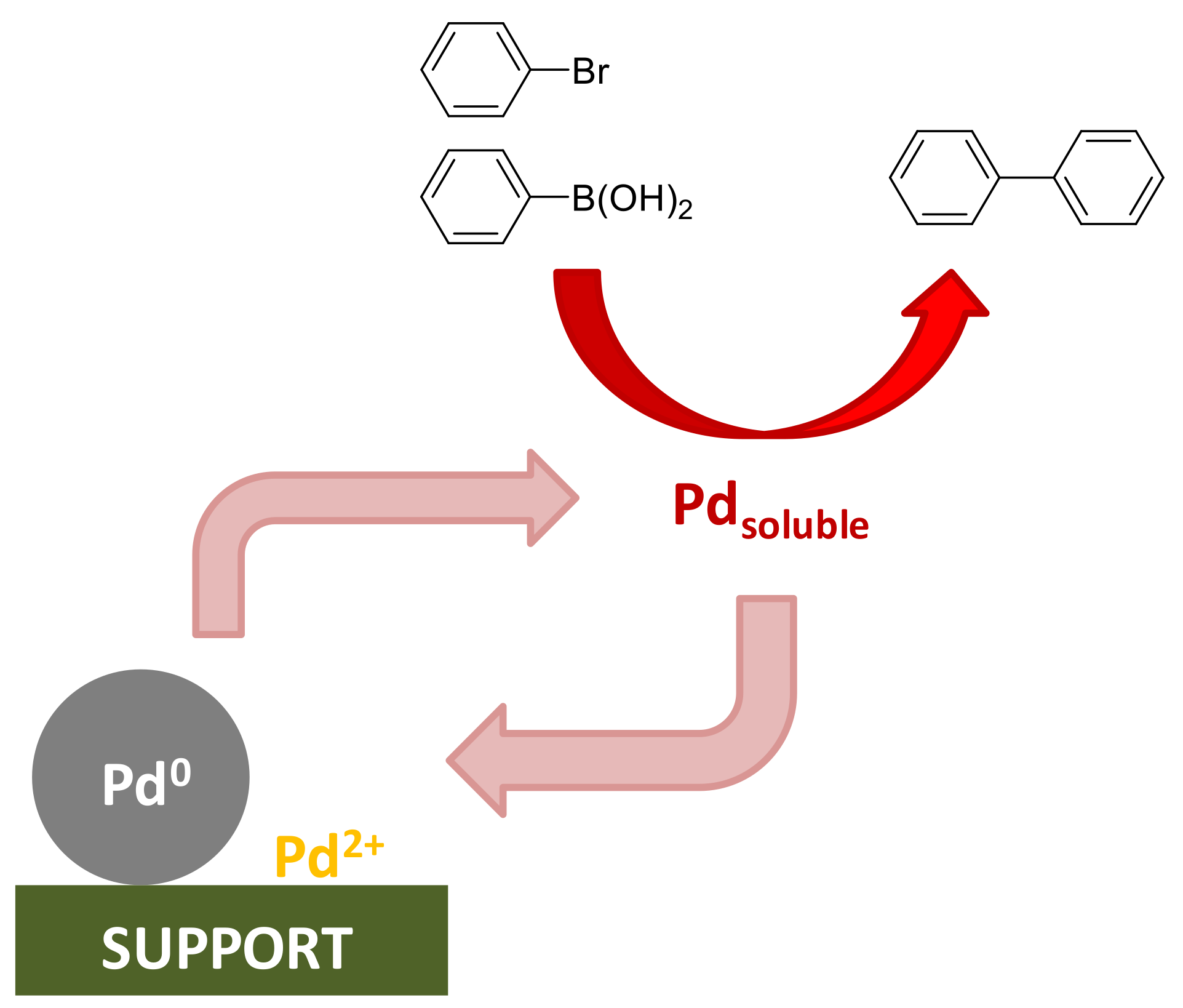
Materials | Free Full-Text | Thiol-Functionalized Ethylene Periodic Mesoporous Organosilica as an Efficient Scavenger for Palladium: Confirming the Homogeneous Character of the Suzuki Reaction

Thiol−Ene Reaction: Synthetic Aspects and Mechanistic Studies of an Anti‐Markovnikov‐Selective Hydrothiolation of Olefins - Sinha - 2019 - Asian Journal of Organic Chemistry - Wiley Online Library

EXAFS Characterization of Pd Catalysts for Enantioselective Hydrogenation of α-Phenylcinnamic Acid: Pretreatment Effects and Thiol Adsorption | SpringerLink

Thiol Treatment Creates Selective Palladium Catalysts for Semihydrogenation of Internal Alkynes - ScienceDirect

KR20140016251A - Palladium-copper catalysts for the homogeneous selective oxidation of thiol groups - Google Patents





