NaBH4-TMEDA and a palladium catalyst as efficient regio- and chemoselective system for the hydrodehalogenation of halogenated heterocycles - ScienceDirect
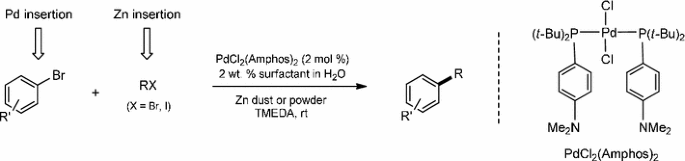
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Pd-catalyzed synthesis of α,β-unsaturated ketones by carbonylation of vinyl triflates and nonaflates - Chemical Communications (RSC Publishing)

Mixed-Ligand Approach to Palladium-Catalyzed Direct Arylation Polymerization: Synthesis of Donor–Acceptor Polymers Containing Unsubstituted Bithiophene Units - Macromolecules - X-MOL

EP2107047A1 - Method for producing organic compounds by means of a transition metal-catalysed cross-coupling reaction of an aryl-X, heteroaryl-X, cycloalkenyl-X or alkenyl-X compound with an alkyl, alkenyl, cycloalkyl or cycloalkenyl halogenide -
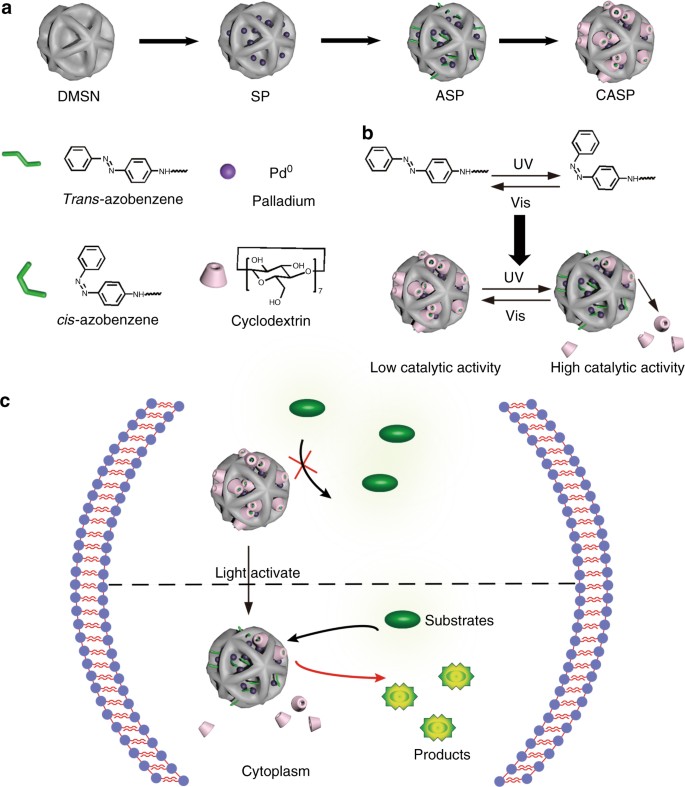
Designed heterogeneous palladium catalysts for reversible light-controlled bioorthogonal catalysis in living cells | Nature Communications

Hydrodehalogenation of halogenated pyridines and quinolines by sodium borohydride/N,N,N′,N′-tetramethylethylenediamine under palladium catalysis - ScienceDirect

One-pot synthesis of self-assembled heteroleptic palladium(II) complexes with tmeda: An application of ligand exchange reactions - ScienceDirect
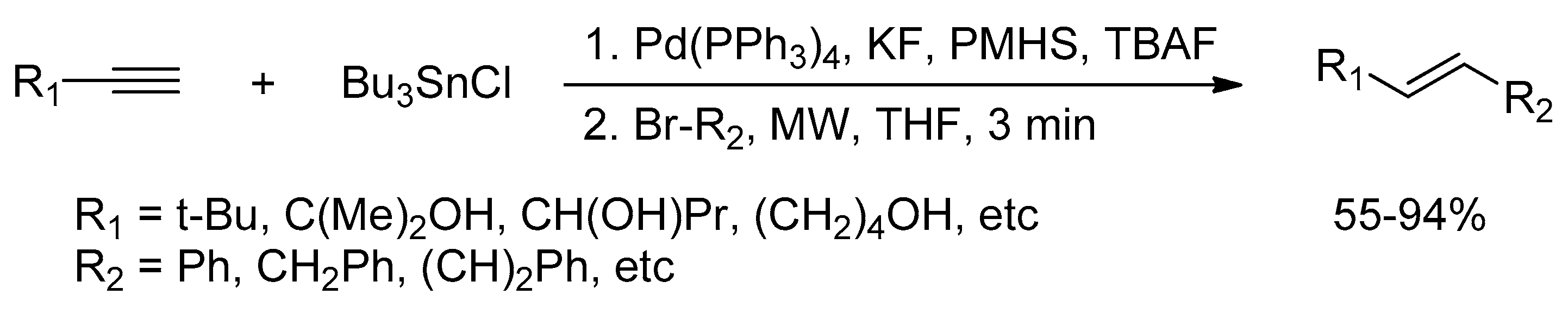
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
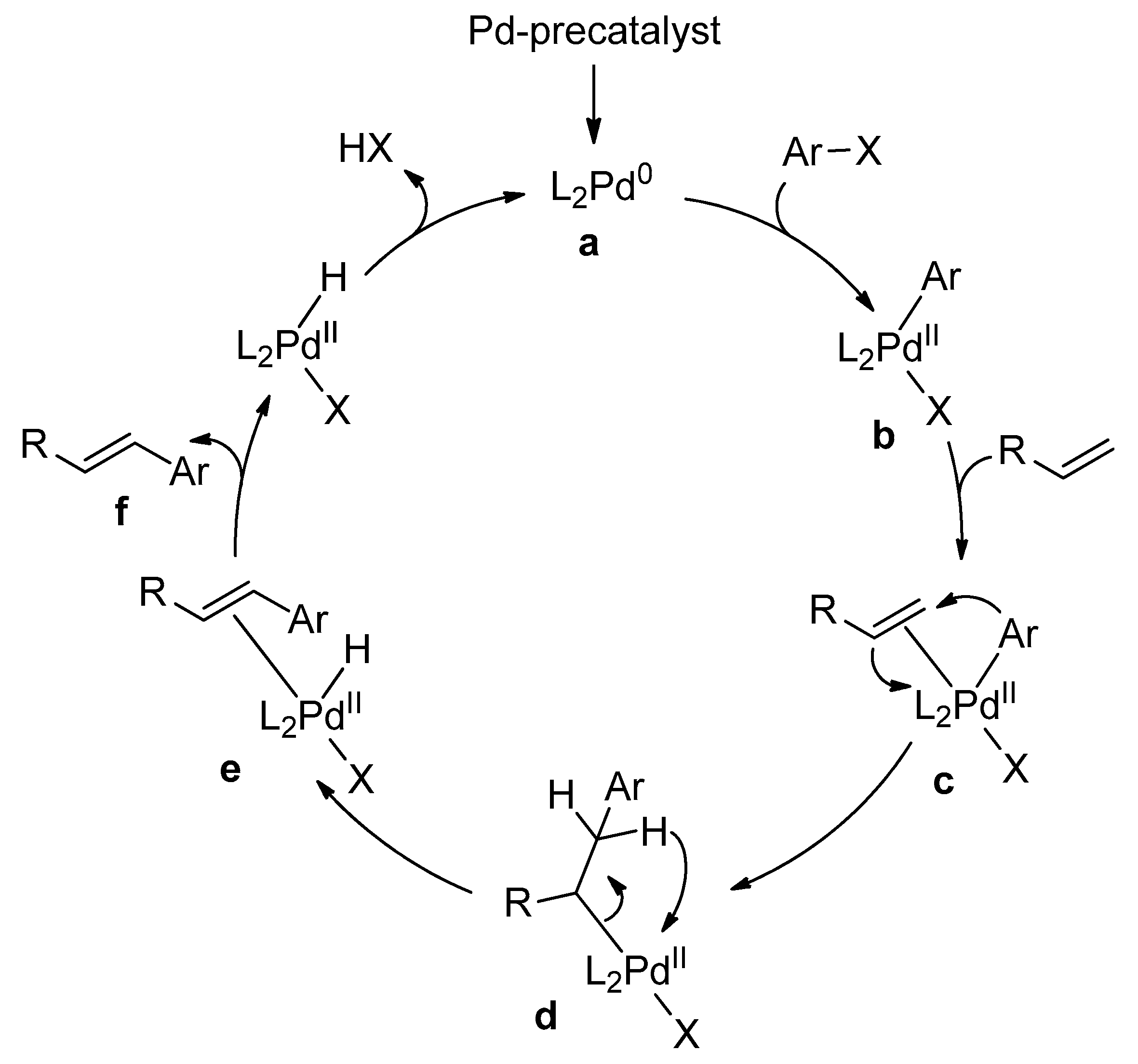
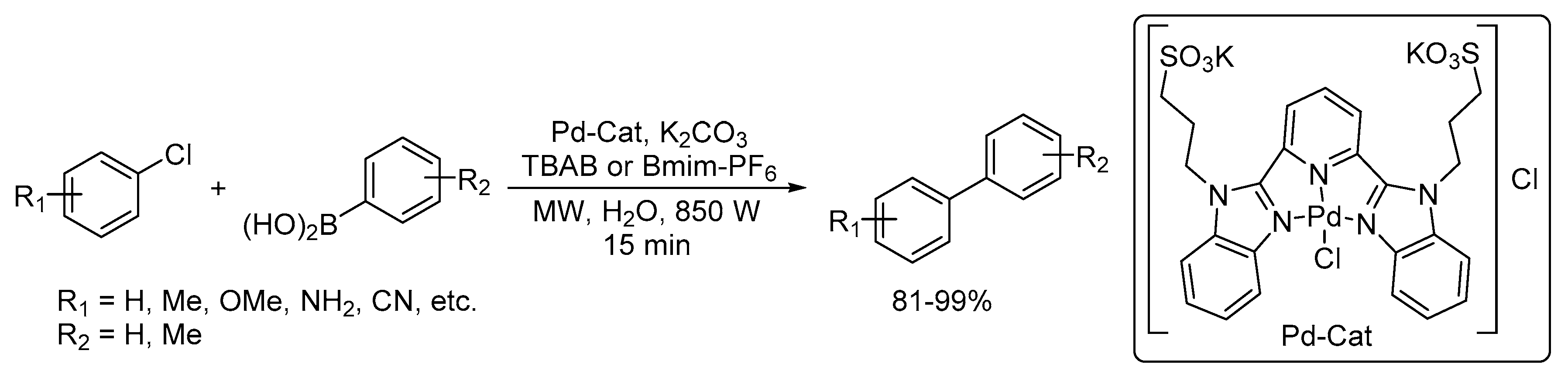
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Iron-catalysed cross-coupling of organolithium compounds with organic halides | Nature Communications
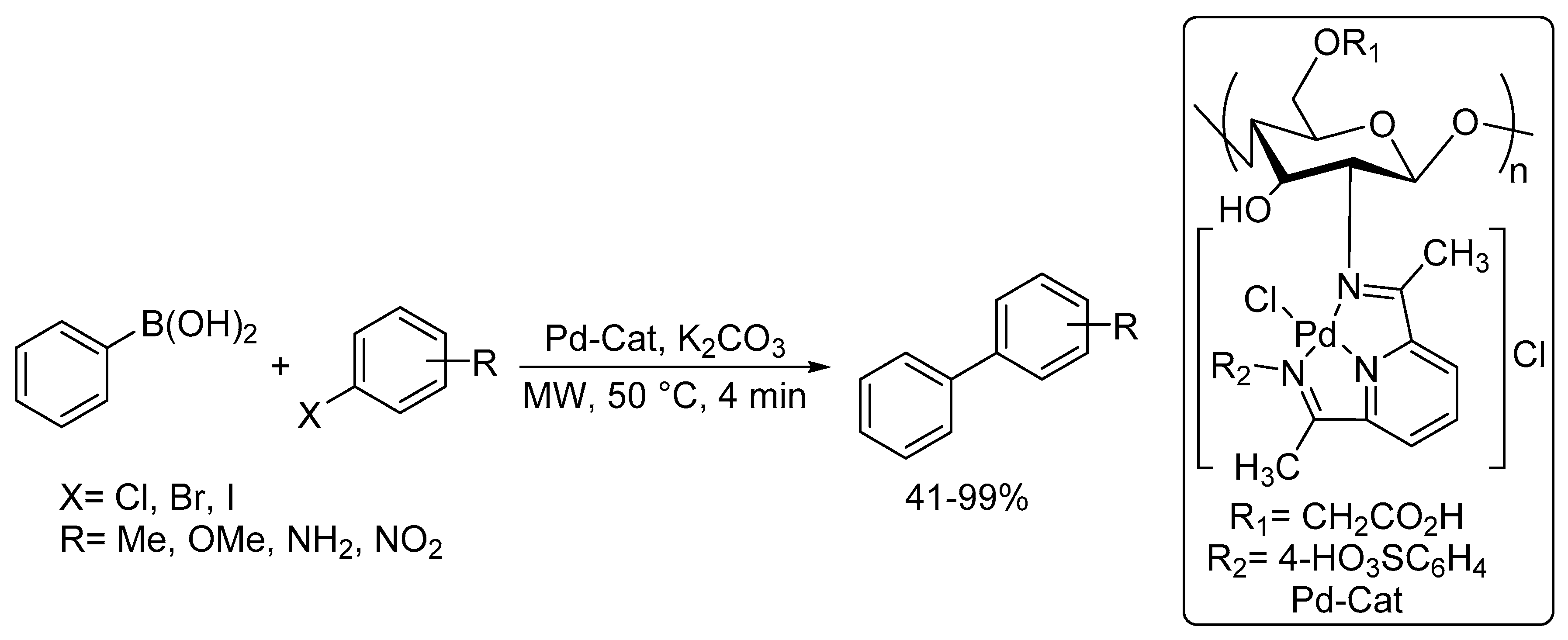
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
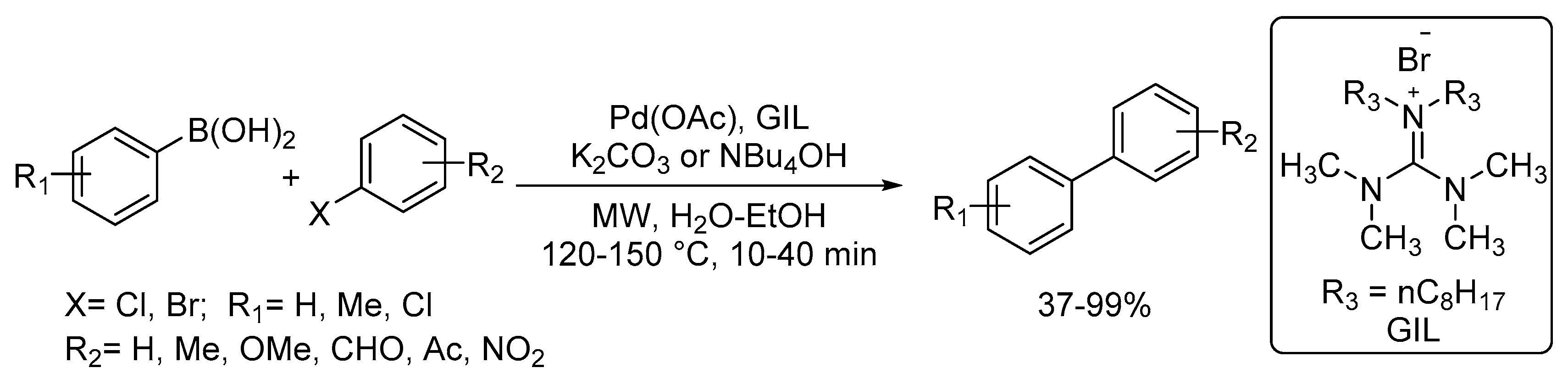
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Hydrodehalogenation of halogenated pyridines and quinolines by sodium borohydride/N,N,N′,N′-tetramethylethylenediamine under palladium catalysis

Synthesis of Internal Alkynes by Pd(PPh3)4/TMEDA‐Catalyzed Kumada Cross‐Coupling of Alkynyl Halides with Grignard Reagents - Zhang - 2014 - European Journal of Organic Chemistry - Wiley Online Library

Regioselective and Stereospecific Cross‐Coupling of Primary Allylic Amines with Boronic Acids and Boronates through Palladium‐Catalyzed CN Bond Cleavage - Li - 2012 - Angewandte Chemie International Edition - Wiley Online Library
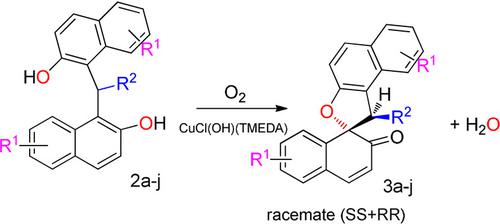
Aerobic Copper Catalytic Oxidation of Methylene and Arylidenebisnaphthols: A Green and Efficient Synthesis of Spironaphthalenones - ChemistrySelect - X-MOL

Optimization of palladium-catalyzed Ferrier-type C-glycosylation with... | Download Scientific Diagram