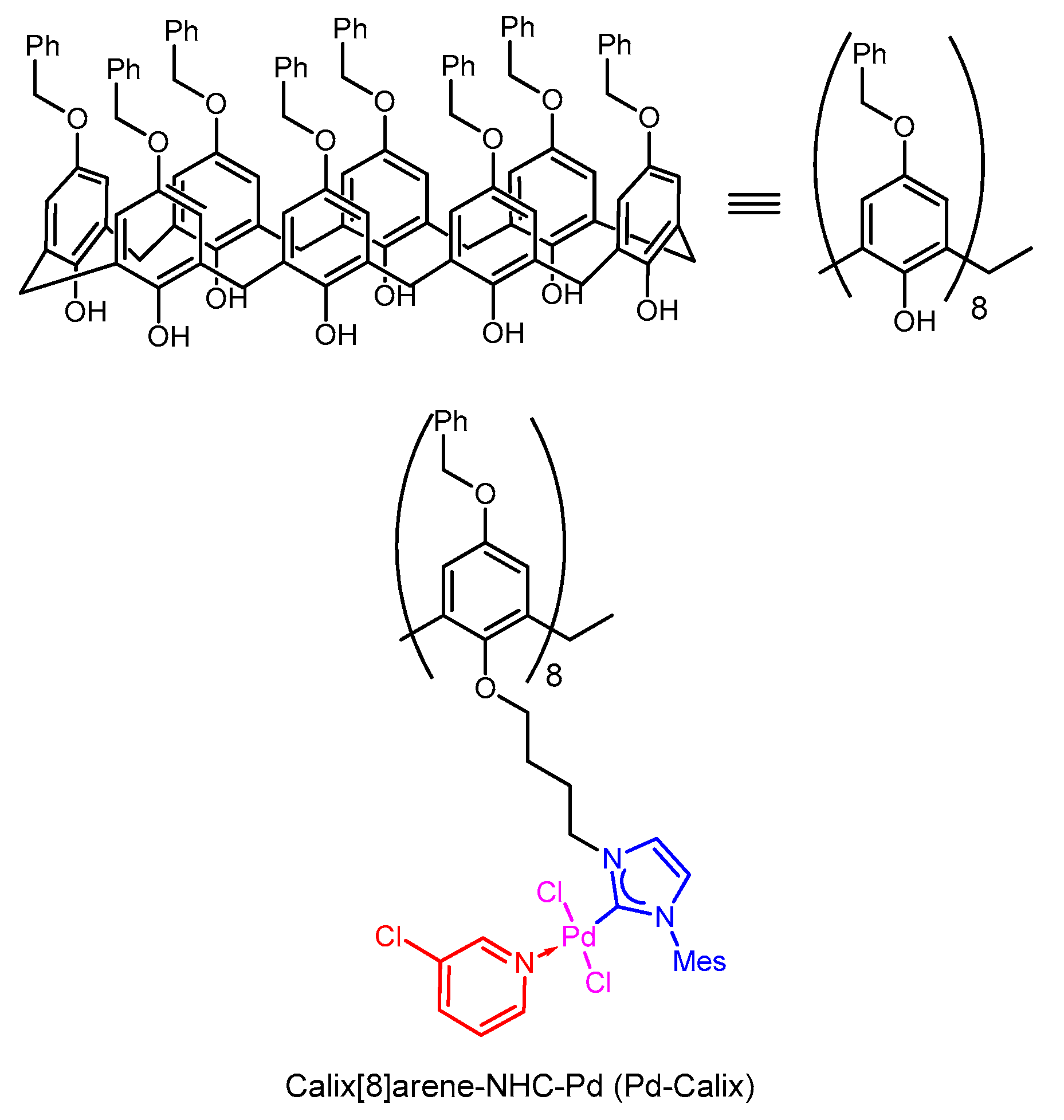
Boronic Acids as Bioorthogonal Probes for Site‐Selective Labeling of Proteins - Akgun - 2018 - Angewandte Chemie International Edition - Wiley Online Library
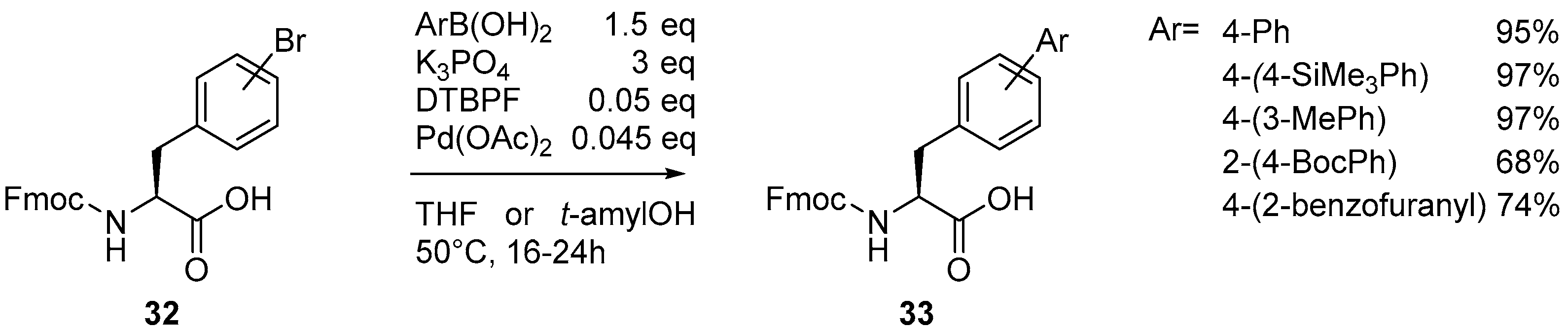
Catalysts | Free Full-Text | The Suzuki–Miyaura Cross-Coupling as a Versatile Tool for Peptide Diversification and Cyclization | HTML
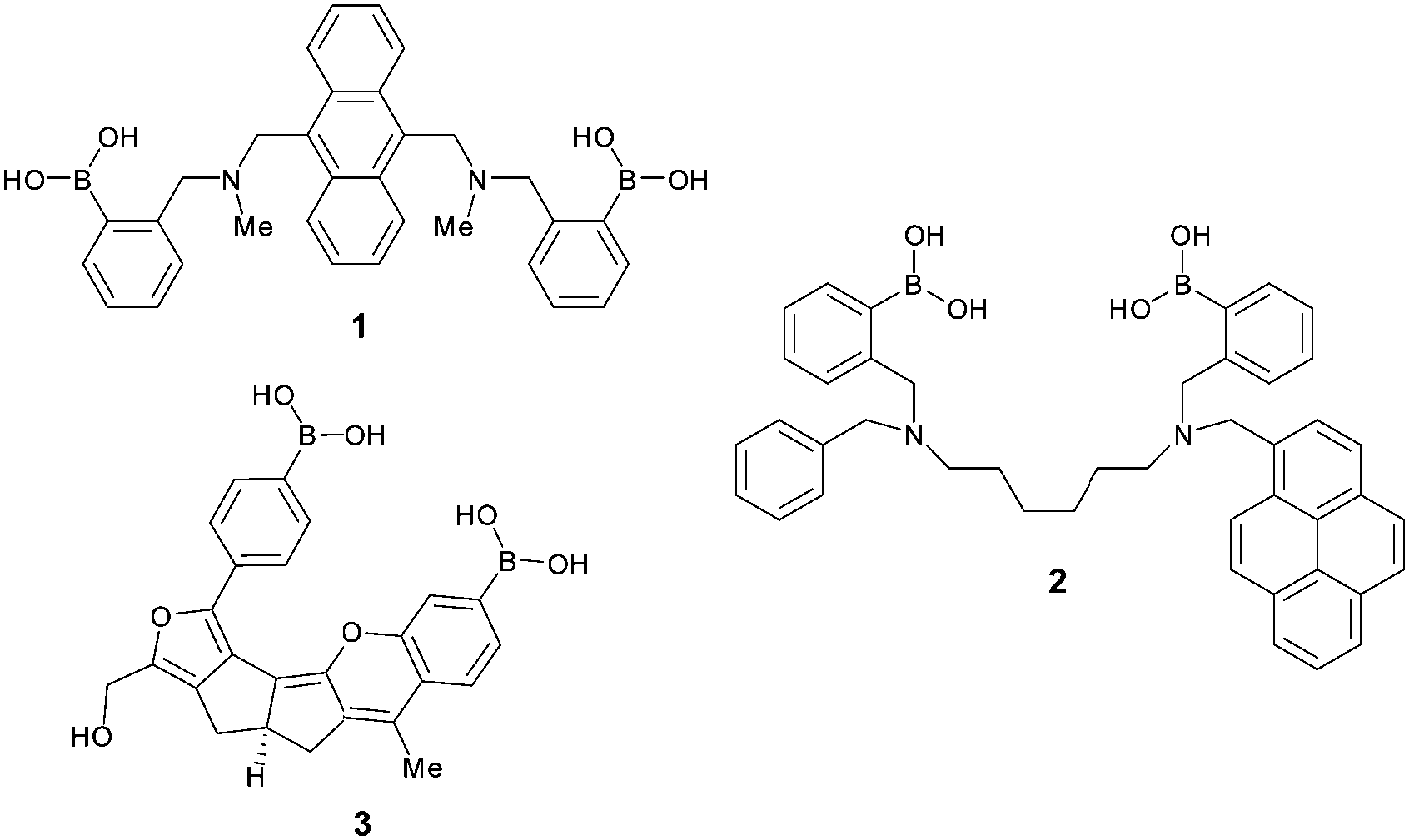
Glucose selective bis-boronic acid click-fluor - Chemical Communications (RSC Publishing) DOI:10.1039/C6CC08534B
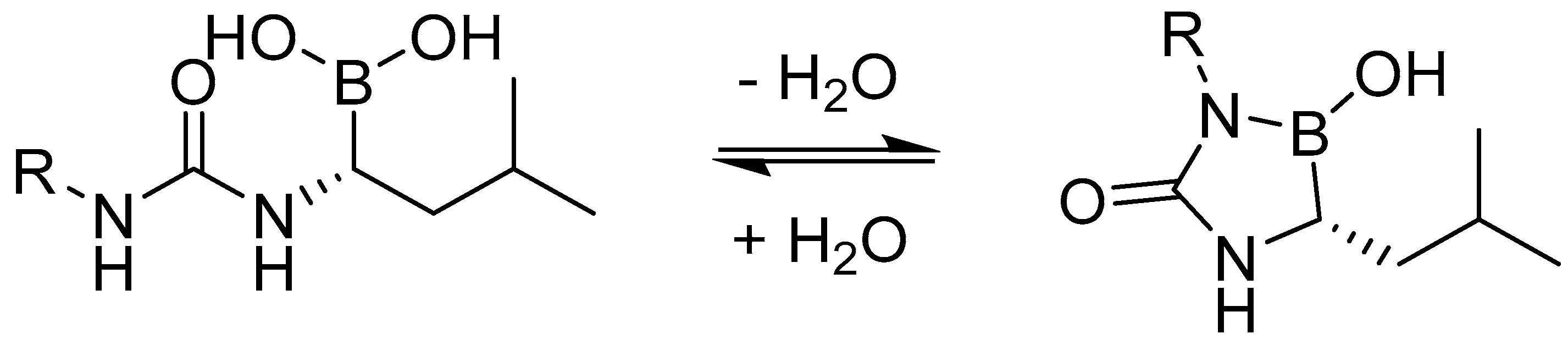
Molecules | Free Full-Text | Boronic Acids and Their Derivatives in Medicinal Chemistry: Synthesis and Biological Applications | HTML
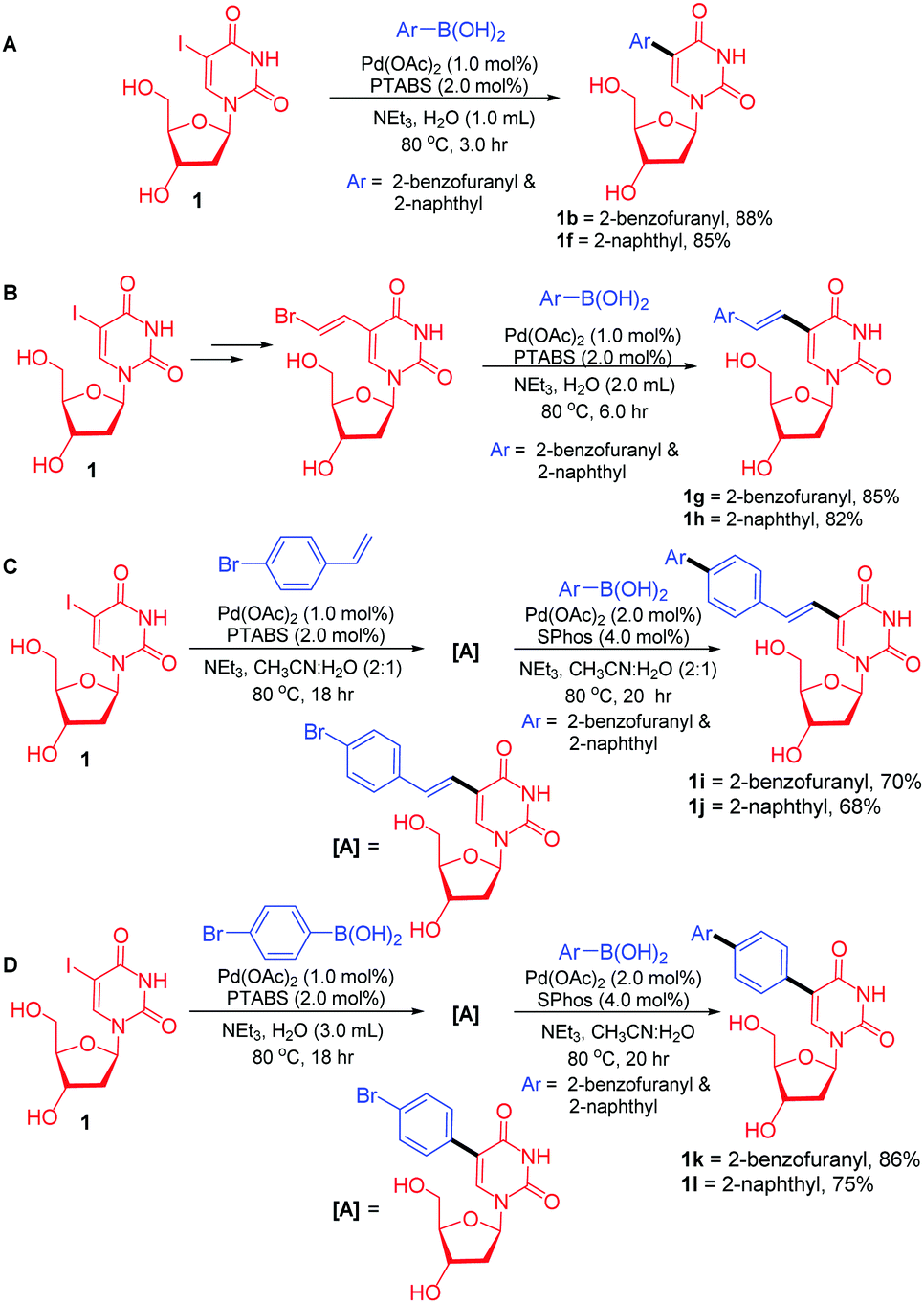
Extended fluorescent uridine analogues: synthesis, photophysical properties and selective interaction with BSA protein - New Journal of Chemistry (RSC Publishing)

Binding equilibria of phenylboronic acid to sugar and OH − ion. OH HO... | Download Scientific Diagram

Cross-coupling reaction on Dha:a)General reaction scheme for the Dha... | Download Scientific Diagram

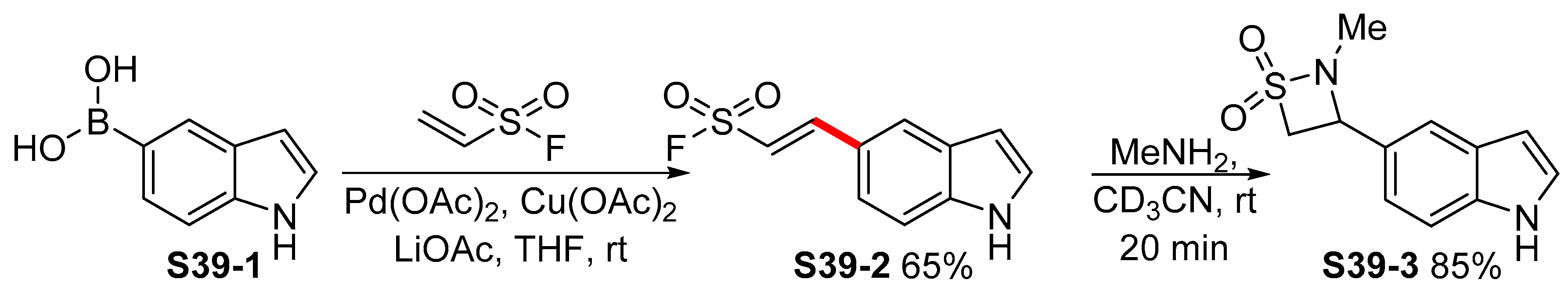
Post-transcriptional labeling by using Suzuki-Miyaura cross-coupling generates functional RNA probes. - Abstract - Europe PMC

Boronic Acids as Bioorthogonal Probes for Site‐Selective Labeling of Proteins - Akgun - 2018 - Angewandte Chemie International Edition - Wiley Online Library

Boronic Acids as Bioorthogonal Probes for Site‐Selective Labeling of Proteins - Akgun - 2018 - Angewandte Chemie International Edition - Wiley Online Library
![Boronic Acids and Derivatives | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation Boronic Acids and Derivatives | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation](https://labchem-wako.fujifilm.com/us/category/images/00002-img02.png)
Boronic Acids and Derivatives | [Synthesis & Materials]Products | Laboratory Chemicals-FUJIFILM Wako Pure Chemical Corporation

A simple and highly selective fluorescent sensor for palladium based on benzofuran-2-boronic acid - ScienceDirect

A highly selective and sensitive boronic acid-based sensor for detecting Pd2+ ion under mild conditions - ScienceDirect
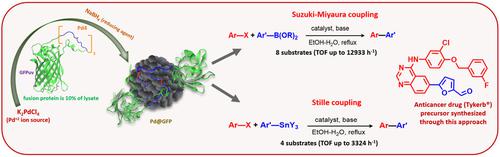
Recombinant Peptide Fusion Protein‐Templated Palladium Nanoparticles for Suzuki‐Miyaura and Stille Coupling Reactions - ChemCatChem - X-MOL